Metals that follow the transition metals can lose their s orbital and p orbital electrons and form charges that are the same as their group number or they can lose their p orbital electrons and keep their two s orbital electrons metals like tin and lead in group 4A can form either +4 or +2 charges. The metals in group 1A like Lithium and Sodium have a charge of +1, the metals in group 2A such as magnesium and calcium have a +2 charge. The main group metals usually form charges that are the same as their group number. Since the metals lie to the left of the periodic table they often have low ionization energies and low electron affinities, meaning they give away electrons relatively easy causing metals to become cations. Finally, metals are good conductors of heat and electricity.Metals are ductile this means they can be rolled into thin sheets.Generally, metals are very malleable, meaning they can be beaten down into thin sheets.Metals are usually more dense than nonmetals.
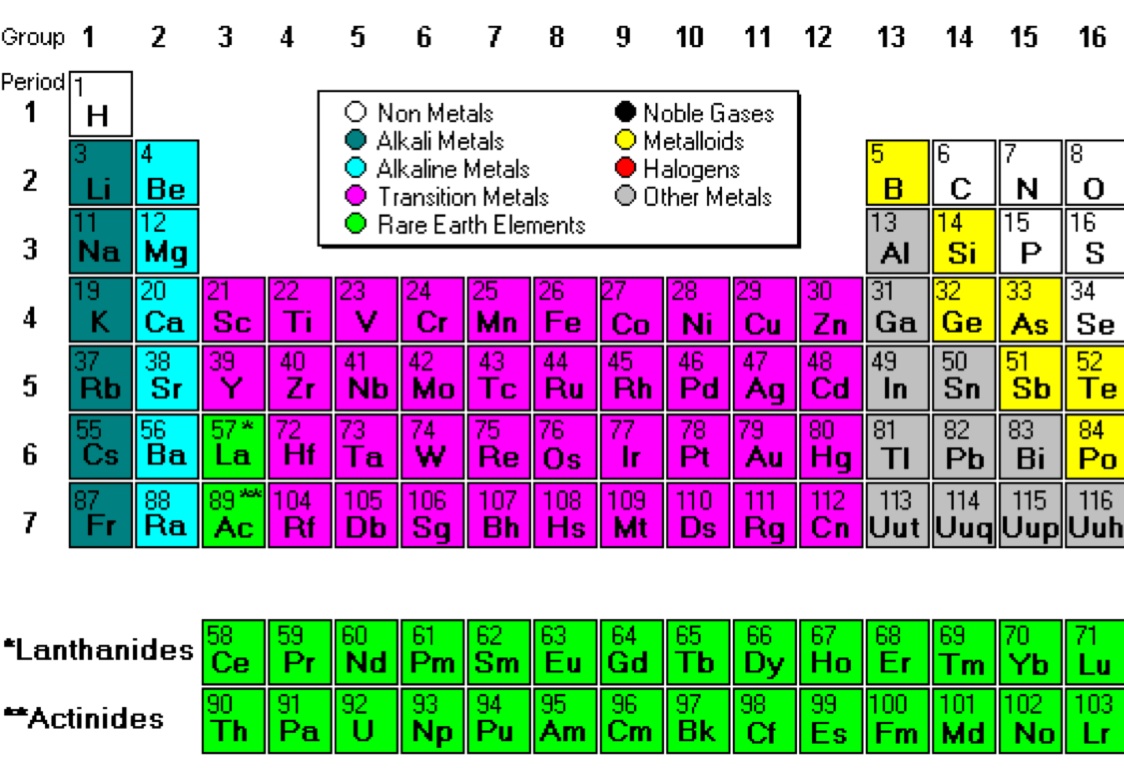
Metals are lustrous in nature, meaning their surface is shiny.Most metals present as solids at room temperature, mercury is the only exception to this rule as it exists as a liquid at room temperature.All metals differ in their melting points, but generally, all metals have high melting and boiling points.That is why jewelry can never be 100% pure gold or else it would not withstand everyday wear and tare. Out of the one hundred and eighteen metals on the periodic table gold is the most malleable. Mercury, which is classified as a metal, is the only metal on the periodic table that presents as a liquid. Some well-known metals on the periodic table include: iron, lead, aluminum, silver, calcium, and sodium which present as solids at room temperature. This means that they have properties of both metals and nonmetals. Elements that happen to fall on the line or just to the left of it (boron, silicon, germanium, arsenic, tellurium, antimony, and polonium) may be classified as metalloids or semimetals. On the periodic table metals, are placed to the left of the zigzag line that runs between the five elements: boron, silicon, arsenic, tellurium, and astatine. Properties of Metals, Nonmetals, and MetalloidsĪ majority of the elements on the periodic table of elements categorize themselves as metals.I hope you have found this article helpful in solving your doubts.įeel free to ask me in the comments, if you have any doubts.In this tutorial, you will be introduced to metals and non-metals on the periodic table of elements and their properties. Then we discussed the total number of metals, Nonmetals and Metalloids present on the Periodic table. In the very beginning of this article, I showed you the Periodic table labeled with metals, Nonmetals and Metalloids. All these known elements are classified in three major categories based on their metallic and nonmetallic characteristics. Periodic table consists of total 118 elements. Get Info + HD Image of Periodic Table Summary You can effortlessly find every single detail about the elements from this single Interactive Periodic table.

Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Why are metalloids also known as semimetals? Free Gift for you: Interactive Periodic Table List of metalloids with atomic number, symbol and names of element.Ģ). I want to keep this article short and I have already explained the reason why astatine and polonium are not classed as metalloids.

There are 6 commonly known Metalloids on the Periodic table.Īstatine and Polonium also shows some characteristics of Metalloids.īut according to some researchers, Astatine and Polonium are not classes as Metalloids. How many Metalloids are on the Periodic table? They are found between the metals and nonmetals.Įlements which have the metallic properties as well as nonmetallic properties are classified as Metalloids.īecause of this reason, they are also known as semimetals. Above picture shows you the labeled Metalloids on the Periodic table.
